Kinetics. Digital image. N.p., n.d. Web. 30 May 2014.
Energy Barriers in Reactions
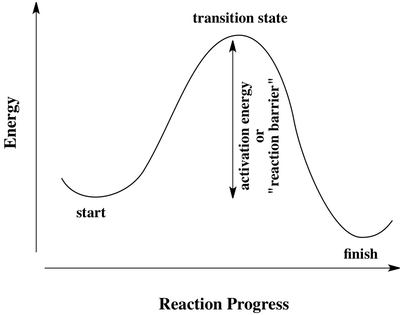
In order for a chemical reaction to occur, the reactants must collide with enough amount of energy to react. With this energy, it's required to achieve the transition state to form the products otherwise without the sufficient energy, the reaction will not occur.
Reaction Profiles
To show how energy of reaction and products during a reactants could be shown in a Reaction Profile. In a reaction profile where there is the highest point is the transition state or the activated complex where energy is at it's highest potential. To trigger into the transition state reactants energy is required to achieve activation energy where the difference between reactants and product is the heat of reaction.
Endothermic & Exothermic Reactions
Endothermic is a reaction that absorbs heat during a reaction process which reactants will lower energy than product.
Exothermic is a reaction that releases heat as the reaction process and the product of it is lower energy than the reactants.
Important Factors that affect Reaction Rate
As we all know,Temperature, Concentration, and Surface Area will influence the outcome of a Reaction Rate and there is a substances called Catalysts which will speed up the reaction by lowering the activation energy. In this process the effect of a catalyst does not change temperature and speeds up both the forward and reverse reactions.
In order for a chemical reaction to occur, the reactants must collide with enough amount of energy to react. With this energy, it's required to achieve the transition state to form the products otherwise without the sufficient energy, the reaction will not occur.
Reaction Profiles
To show how energy of reaction and products during a reactants could be shown in a Reaction Profile. In a reaction profile where there is the highest point is the transition state or the activated complex where energy is at it's highest potential. To trigger into the transition state reactants energy is required to achieve activation energy where the difference between reactants and product is the heat of reaction.
Endothermic & Exothermic Reactions
Endothermic is a reaction that absorbs heat during a reaction process which reactants will lower energy than product.
Exothermic is a reaction that releases heat as the reaction process and the product of it is lower energy than the reactants.
Important Factors that affect Reaction Rate
As we all know,Temperature, Concentration, and Surface Area will influence the outcome of a Reaction Rate and there is a substances called Catalysts which will speed up the reaction by lowering the activation energy. In this process the effect of a catalyst does not change temperature and speeds up both the forward and reverse reactions.